Calcium Imaging
CALCIUM IMAGING: FUNCTIONAL IMAGING
WHAT IS CALCIUM IMAGING?
Calcium plays a key role in cell signaling: the movement of Calcium ions provoque a change in cells membrane potential, which is partly responsible for neuronal signals transmission. Calcium signaling regulates a wide range of physiological properties of cells.
Calcium Imaging is an essential technique to evaluate Calcium ions role in functioning neurons. The Calcium Imaging technique uses a fluorescent “dye” that responds to the binding of Ca+ ions and changes its fluorescent properties, allowing optical measurement of Calcium concentration shift within neurons and neuronal tissue.

Thanks to Calcium Imaging, you can either measure individual cell or global neuronal network Calcium dynamics at a very high resolution. Calcium Imaging analysis tools allow to circle regions of interest in the studied neuronal culture, so that AP firing can be measured locally.
Calcium imaging is a mid-throughput technique.
WHAT FOR?
We can perform Calcium Imaging on:
- Cloned channels and receptors
- Rodent brain neuron cultures
- Rodent brain slices
- Human iPSC-Derived neurons
- Rodent DRG sensory neurons cultures.
Calcium imaging on rodent DRG sensory neurons can help investigate at the earlier stages of the drug discovery process the pharmacology of compounds developed for Pain pathologies. If you want to learn more about how we can provide you with a complementary study on human DRG sensory neurons, please visit www.neuroservices-alliance.com.
ADVANTAGES OF CALCIUM IMAGING
NEURONAL CULTURES RECORDINGS
Calcium imaging helps document and understand compounds activity at the single neuron level or neuronal network level.
Measurements
- Firing activity of small neuronal subpopulations
Case study: Capsaicin-induced firing activity
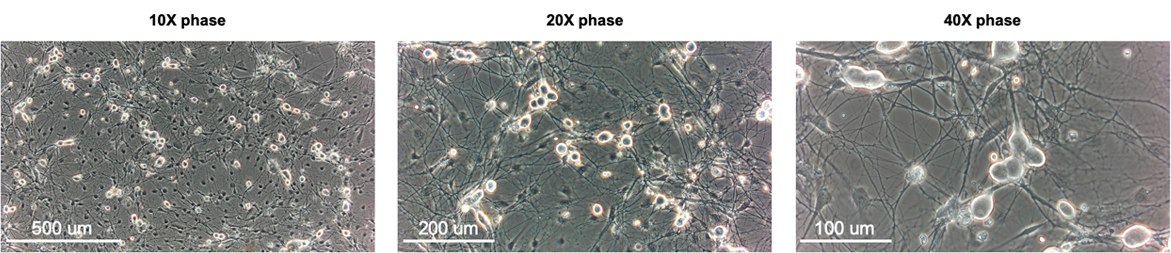
Sensory neuron cultures prepared from adult rat dorsal root ganglia and plated on a monolayer of astrocytes growing on PDL-coated glass imaging dishes (MatTek). Ca imaging was performed on Fura-2 AM loaded cells at 9 days in vitro.
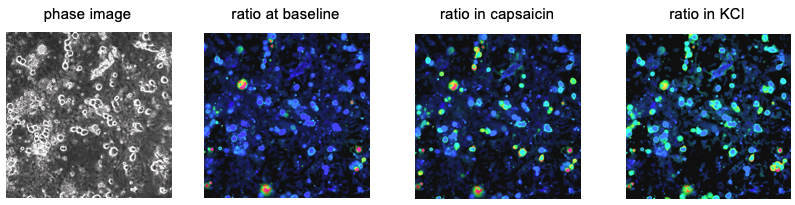 Phase-contrast (left) and ratiometric images of Fura-2 loaded DRG sensory neurons. 300nM capsaicin (TrpV1 agonist) elicited calcium responses in ~50% of sensory neurons.
Phase-contrast (left) and ratiometric images of Fura-2 loaded DRG sensory neurons. 300nM capsaicin (TrpV1 agonist) elicited calcium responses in ~50% of sensory neurons.
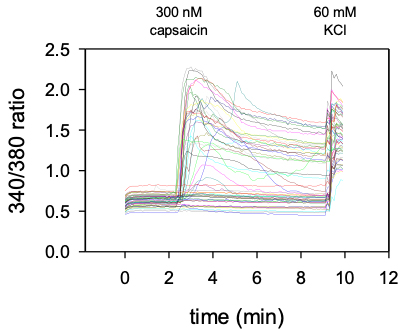
Time course of calcium responses in individual Fura-2AM loaded DRG sensory neurons indicated by colored lines.
Following a 2 min baseline period, perfusion for 1 min with 300 nM capsaicin (TrpV1 agonist) elicited calcium responses in ~50% of neurons expressing TrpV1. All cells responded to depolarization by 60 mM KCl indicating healthy, electrically excitable neurons.
Rat cortex neuron cultures
Neuron cultures were prepared from E18 rat cortex and plated on a monolayer of astrocytes growing on PDL-coated glass imaging dishes (MatTek). Calcium imaging was performed in Fura-2 AM loaded cells at 9 days in vitro.
Perfusion with 100 uM glutamate elicited an influx of calcium in E18 pyramidal neurons.

