News
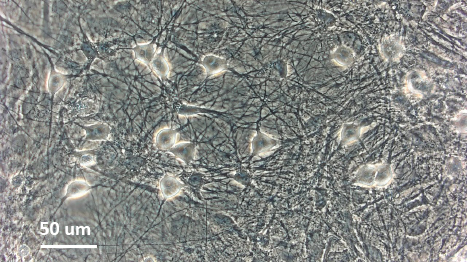
Human iPSC-derived sensory neurons for pain drug discovery
Drug discovery workflows for neurological indications have traditionally relied on neuronal preparations from rodent tissues. However, the ability to test activity of compounds on human neurons is desirable, since rodent pharmacology data do not always predict effects in humans. Human iPSC-derived neurons are increasingly used to provide human in vitro pharmacology data, with the promise of improving decision-making for neuroscience drug discovery. In pain research, human iPSC derived sensory neurons have been used by recent studies to model primary sensory neurons of dorsal root ganglia. Although different types of human iPSC-derived neurons have been extensively characterized by gene expression, the most salient phenotype of neurons is electrical excitability, as neurons integrate synaptic inputs and respond by firing action potentials.


Photomicrographs of Axol iPSC-derived sensory neurons (left) and adult dorsal root ganglion DRG neurons (right)
Patch-clamp electrophysiology assays are routinely used to characterize neuronal excitability and provide functional endpoints to assess effects of drug compounds. However, the basic electrophysiological properties of human iPSC-derived sensory neurons have not been well studied.
How well do iPSC-derived sensory neurons reflect the physiological state of native primary sensory neurons? How consistent is the phenotype? How reproducible are the data?
To address these questions, we focused on human sensory neurons differentiated from iPSC-derived neural progenitors (Axol Bioscience, ax0055). We conducted a thorough patch-clamp analysis, including:
1) Current-clamp recordings to measure spontaneous and evoked action potential firing
2) Voltage-clamp recording to characterize tetrodotoxin-sensitive and tetrodotoxin-resistant Na currents (found in native neurons).
We also determined the concentration-response of tetracaine, a voltage- and frequency-dependent blocker of voltage gated Na currents. We recorded >500 neurons from twelve independent neural precursor differentiation experiments.
We conclude that human iPSC-derived sensory neurons reproduce several key electrophysiological properties expected from bona fide dorsal root ganglia sensory neurons and are useful for supporting drug discovery programs for pain.

